I'm trying to fix the battery pack for an old 486 laptop. If I understand correctly, this was the first on the market with Li-Ion (as well as TFT!).
I replaced the original 18650's a couple years back, then let it sit for a year or so in storage... And sure-enough one of the two pairs of year-old cells was completely kaput.
So this time I looked at the circuitry in the pack to see what could've happened... and... well, I think I have a vague-ish idea.
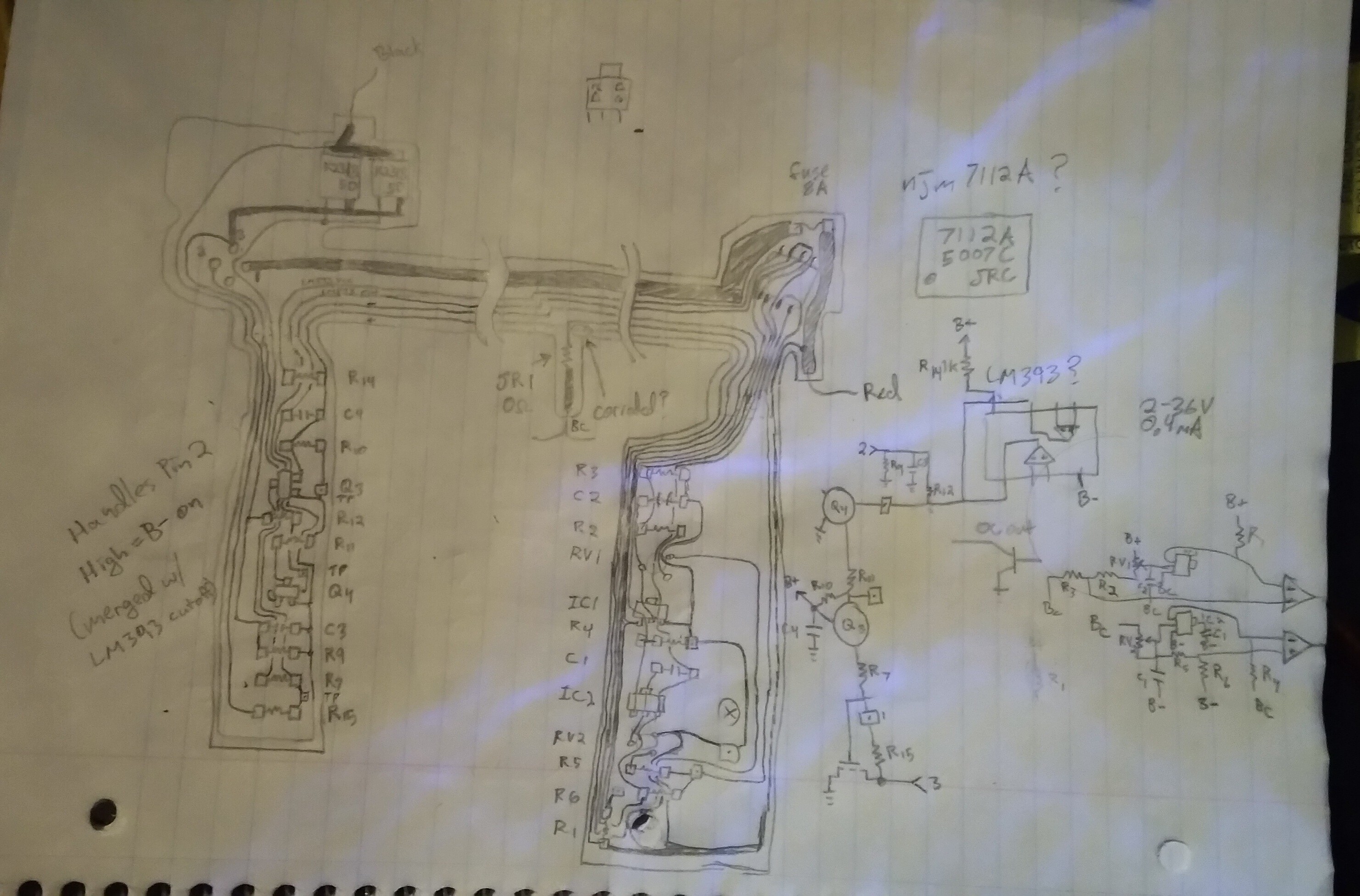
It would seem this thing's protection circuitry is very limited compared to today's.
An actual fuse takes care of over-current. But otherwise it seems all that circuitry does only two things: cut off discharging when one of the parallel-pairs in the series is too low, and cut off output when it's not in the laptop (don't want it shorting on a paperclip!).
Unless I'm missing something *that's all* it does.
No charge balancing, despite its having a wire on each cell-pack.
No thermal cutoffs.
In fact, nothing to prevent it from charging (since the cut-off MOSFETs have body-diodes).
I dunno what all.
Funny thing is, the connector has four pins. Only three are used.
Basically the entirety of this circuitry could be in the laptop itself, excluding the paperclip-fire-preventing transistors. Their gate could be on one of those pins... along with another pin for measuring in the middle of the series.
And then...
Well... I think this would've prevented what I guess must've ruined one of the parallel-cell-pairs:
The over discharge circuit is *always* powered. From the entire series.
Meaning:
A) I probably forgot to charge it before storing, so the cells were already low.
So, after a year of the LM393 comparator sipping 0.4mA, the pack was just *empty*
B) I used cells that were not brand new, nor had been used the same number or depth of cycles.
So, as the series cells discharged, one pair reached zero before the other, then began charging backwards from the other.
...
Which most cell chemistries do not like. Heh.
But the reason this intrigues me is that allegedly Li-Ions are basically unchargeable if allowed to drop too low, something like 1V. Yet I've been running experiments on many old packs and finding, repeatedly, that they definitely can be recharged even if they've been sitting for years and measure less than a diode-drop.
So, it makes me wonder about the thought-process at the early-days of the Li-Ion...
NiCd cells, for instance, are known to be fine even if stored with their terminals shorted for years. In fact, I've heard that if you're planning to store them, then that's the best way.
I wonder if that's basically what the designers of my 486 laptop were thinking... (though why would they bother with a low-voltage cutoff circuit? maybe that's not what it is?)
BUT that logic only works if they're Not In Series. Because, if you have one cell at 0.8V and another at 0.78, and put a resistor between to drain them, then eventually you'll have one cell at 0.01 and the other at -0.01... and that negative charge is what does a cell in. As I Understand.
...
I dunno this laptop is pretty decent quality, and wasn't cheap when it was new, so I can't quite picture they were cutting corners.
...
Anyhow, again, I did manage to recover one of the pairs, even though it was at something like 0.15V. The other, though... Nothing doing. For a moment I got it up to 0.8V, but then despite pumping current in its voltage began to drop.
I'm no expert, so I figure that's time to call it.
..
whoa, if I'm reading my notes right, the surviving cells are holding nearly the same charge as a regular pack of the same make of cells with its "proper" protection circuitry that never goes lower than 2V. (?!)
...
In other packs...
Well... I've looked at one from an iBook G3, and another from a pentium 150 (I think)... both had been sitting for years and definitely way below 1V per cell.
MOST of the cells definitely have useable capacity left in them... Though, definitely diminished from their "new" capacity.
Also a tremendously larger output resistance.
But, still plausibly useful, somehow.
I keep thinking: these were basically useless, as I recall, giving about 5minutes runtime on the laptops.
I ran experiments on single cells, loading them with 1A, and they cut-out at about 3-5min, with the "proper" under-voltage protection.
But, when I parallelled those same two cells, I got 45min, at 1A.
... TEN-FOLD...
I imagine several factors at play, here...
Basically increased output resistance and the low-voltage cut-off. But a few others a bit more vague in my mind... Something about "surface charge" maybe? E.G. after the load is removed, after the low-voltage cut-off, the cells measure around 3V, but after about five minutes they measure more like 3.8(!) [allegedly that's a "full charge!"].
...
I haven't really figured out how to make use of these cells. To get the most out of them, I think, a really sophisticated load-balancing scheme would be ideal...
But, I should point out that Li-Ion cells can be dangerous, and I really have no idea whether it's a good idea to continue using them like this, when they're definitely way past their prime, but still holding a useful-enough charge for repuposing.
...
Though one thought keeps coming to mind: E.G. if you've got an old phone with a couple old batteries that barely even run long-enough to boot without being plugged-in... *paralleling* them might be the difference between not being able to make even one call and its working fine for possibly several long-winded calls.
But, again, consider possible risks, here... Doing-so with two decent batteries (with low output resistance) could send a LOT of current from one to the other, even if their voltages are near identical.
 Eric Hertz
Eric Hertz
Discussions
Become a Hackaday.io Member
Create an account to leave a comment. Already have an account? Log In.