Early on I figured I'd need to test some CR2477 cells to see how much energy they could practically supply in a short time. I finally got around to building the tester and putting it to work (not to spoil the surprise, but things don't look great so far). Here's the tester:

The circuit is basically what I described in an earlier log, except with a few R's and a C added for stability.
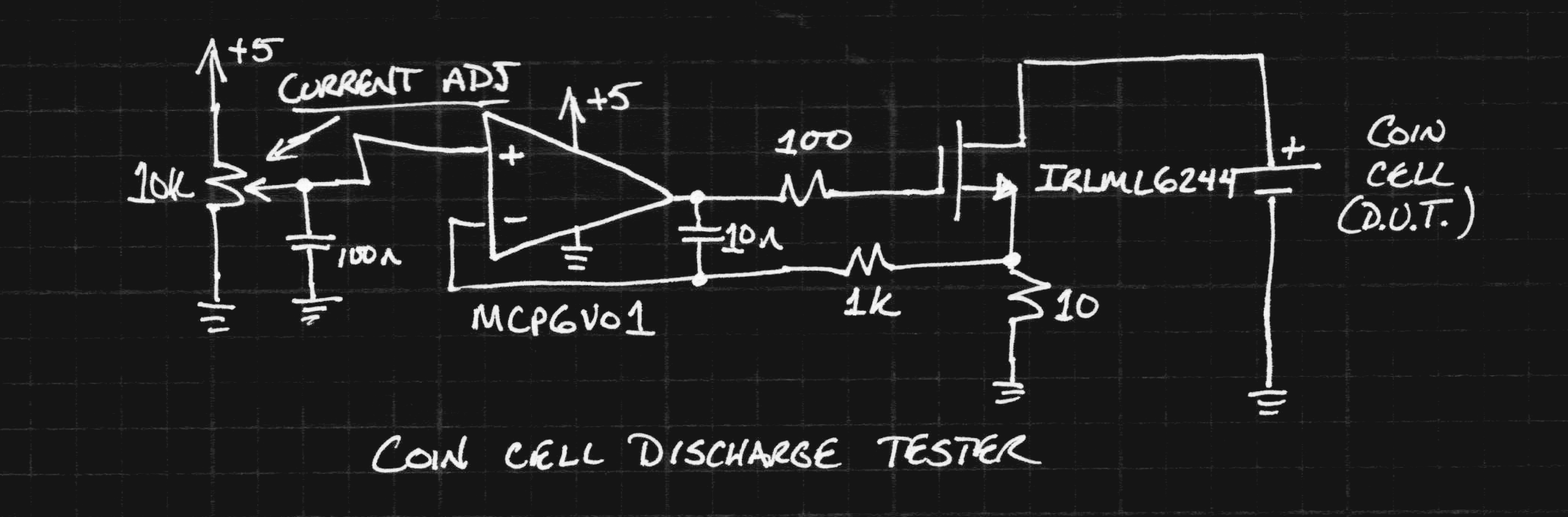
The first low-offset op-amp I found in the parts bin was an MCP6V01 chopper. It's overkill for this application but contributes a ridiculously small error to the result. The cell is discharged at a constant current set by the potentiometer, while serial-port-enabled DMMs log the current and cell voltage (not shown). I didn't have any 1% 10-ohm resistors with a decent power rating, so I paralleled 10x 100-ohms. The MOSFET and op-amp are mounted on #Ugly SMD Adapters.
My initial plan was to discharge the CR2477 at a constant 40mA. This represents a C/25 discharge rate, which is completely reasonable for any battery other than a coin cell. For the CR2477, it turns out, this is an enormous drain that wastes most of the energy in the cell. Here is a plot of the voltage during the 40mA discharge as well as the total energy (in this case dissipated as heat in the 10-ohm resistor):
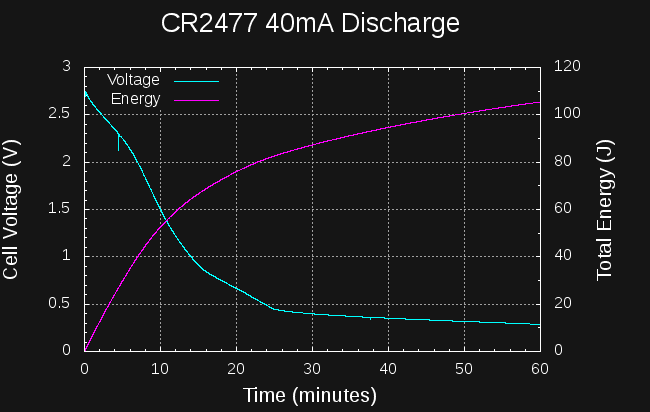
After 10 minutes of discharge, the cell voltage has decreased below 1.5V, which I think is as low as I can reasonably expect to go using a PIC12LF1571 in the DC-DC converter (the PIC is specified down to 1.8, but I will push it). At the 10-minute mark, the energy extracted from the cell is around 55J. Terrible. Integrating the current to this point yields 6.6 mAh, which is 0.66% of the cells capacity. Awful.
I let the drain continue for about 9 hours, even though after 25 minutes, the cell voltage dropped below the 400mV required to maintain a 40mA discharge in the load resistor. At the end, 157J had been extracted. Ghastly.
Lesson: don't discharge CR2477 cells at 40mA.
What about 20 mA?
Undaunted, I discharged a CR2477 cell at 20mA (note the different time scale).
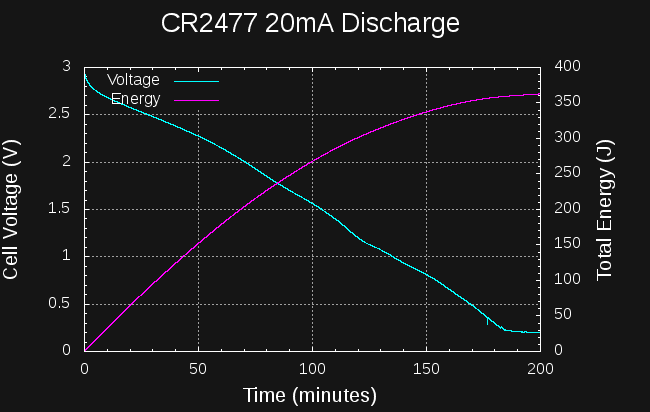
In this case, around 260J and 35 mAh have been extracted from the cell before the voltage drops below 1.5V (lousy). This 5x as much as the 40 mA discharge, but still represents a small fraction (3.5%) of the capacity of the cell. Atrocious. When I finally stopped this test after about 8 hours, 392 J had been extracted from the cell. Dreadful.
Lesson: don't discharge CR2477 cells at 20mA.
Are you seeing the pattern yet? Lower currents will be required to extract more energy from the cell. Of course, these discharge curves don't come anywhere near the 25-hour estimate I originally used for charging (the 20mA discharge is essentially done in 3 hours). Still, the total amount of energy I have been able to obtain from the cells is very low. Abysmal.
What I Got Wrong
So, in my initial estimates of current drain, I assumed that a 1000mAh CR2477 was like a 225mAh CR2032 scaled up by 4x, so I could draw 4x the current (I found data fro CR2032s on the web). The differences between the cells are more subtle than that. The maximum current you can draw is proportional to the electrode area, and assuming the electrode size is proportional to the cell diameter, a CR2477 only has 24^2/20^2 = 1.44x the electrode area. The total amount of reactive chemicals inside may be 4x, but the maximum current you can reasonably draw is likely to be around 1.4x.
I also based my initial estimates on numbers in this article, which says you can use 88% of the available energy in a CR2032 when draining it at 10 mA. Unless CR2032s are much better at higher currents than CR2477s, I just don't see it. I should test a CR2032 for comparison.
What's Next?
There is a trade-off between the length of time taken to drain the cell and the self-discharge of the supercapacitors. The longer you take to drain the cell, the more energy you get, while the more you lose due to self-discharge in the cap. Somewhere in-between, there is a maximum amount of energy you can get into the capacitor.
I'm currently running two more tests. First, discharging a CR2477 cell at 10mA. So far, this is looking better, already having extracted 845 J in about 10 hours, and the voltage is still about 1.5V.
The second test is to get a better estimate of the self-discharge of the 400F supercapacitors. I charged one to 2.33V, and am logging the voltage as it self-discharges. At first, the selft-discharge was around 5.5mA, which is absolutely terrible, but it is dropping steadily now. This is probably due to some of the charge being a surface or shallow charge in the capacitor.
Data from these tests should give an indication of the optimum duration for charging the capacitor from the cell, and how much energy can be expected.
Bootstrapping the PIC
One more idea occurs to me. It seems that under "heavy" drains like these, there can still be a significant amount of energy left in the cell even when the voltage drops to 1.5V or lower. So, why not have the PIC bootstrap its own power supply? Once the supercapacitor is charged to more than the cell voltage, the PIC can be run from the output voltage (a pair of BAT54 Schottky diodes can switch the PICs Vdd). A low-quiescent regulator might be necessary, but the scheme should work, and will allow more energy to be drained from the cell.
Bootstrapping also gives me an idea for running TritiLEDs from single AA cells (lithium or otherwise)...
 Ted Yapo
Ted Yapo
Discussions
Become a Hackaday.io Member
Create an account to leave a comment. Already have an account? Log In.
Are you sure? yes | no
I didn't consider bootstrapping initially because all the manufacturers say coin cells are dead at 2.0V, so I figured there was no need to run anything below that. But draining them this quickly is definitely an "off label" use, so the 2.0V limit doesn't necessarily apply. It just took a little while to connect the dots.
I essentially have a two-step charging process. The ceramic caps on the board average out the current from the cell - the inductor draws several hundred mA ramps of current, but the cell sees a much lower average rate.
But, if I could find large enough capacitors with low enough self-discharge rates, I could charge these intermediate caps over a week or so, then use those to charge the supercaps much more quickly.
Wait! Maybe you hit the solution here. Fucking brilliant. What about starting with a pair of discharged LSD NiMH cells, and using that as an intermediate reservoir. You could take a week to charge the NiMH battery, getting good capacity from the coin cell, then charge the supercap in just a few hours from the NiMH cell. You'd have to measure carefully, and convince everyone that you aren't using any energy that was already in the NiMH battery, but it should work.
The problem is that state-of-charge is difficult to measure accurately in NiMH cells, and discharging them all the way down to 0 damages them. I know you can store NiCd cells shorted without damage. Maybe charging a pair of previously shorted AA NiCd cells from the coin cell over a week, then charging the supercap from that. Ha!
I mean what's the difference between storing the coin cell energy temporarily in an electrochemical capacitor or an electrochemical secondary cell?
Now I won't be able to sleep tonight...
Are you sure? yes | no
Are you sure? yes | no
Are you sure? yes | no
Something seems wrong with the numbers you're getting; have you seen this white paper? http://www.ti.com/lit/wp/swra349/swra349.pdf
Are you sure? yes | no
I wouldn't be too surprised if there were something wrong with my numbers - I'd actually be a little relieved - but the TI paper is about a very different use case.
I read it before. They are testing very low duty cycle pulses of 30 mA. Their drain profile (on page 3) is 8mA * 2ms, 30mA * 1ms, 8 mA * 2ms, 0.1mA * 200 ms, so the average current is only 0.4 mA. My 40mA test was 100x this average current.
It's no surprise that the cells can supply short bursts of high current, but in order to charge a supercapacitor before the caps self-discharge bleeds off all the energy, you have to somehow manage a high average current. This has thus far proved elusive.
Are you sure? yes | no
I took a look at CR2477 datasheet from Sony https://www.sony.net/Products/MicroBattery/cr/pdf/cr2477.pdf and they state quite steep decrease in capacity at loads above roughly 1mA
picture here-> snag.gy/5QEN84.jpg
Honestly speaking, it's quite concerning for me too. I took a look at Panasonic and saw similar
picture here-> snag.gy/QRAgjT.jpg
Notice the right graph, it's for CR3032 (not 2032). Approximately one order of magnitude more beefy than CR2477 (left graph). Here is comparison of CR2032 and CR2330
picture here-> snag.gy/CgGd5U.jpg
It looks to me that at higher currents (anything above few miliamps) the actual performance of CR2477 falls below CR2032.
Are you sure? yes | no
Yes, things are not looking good.
It's almost like the CR2477 sacrificed high current capability for high capacity. I did not expect that. Once the CR2477 in the jig is done testing, I'll throw in a CR2032 at the same current and see what happens.
Then there's the temperature dependence. Maybe there's something to be gained - the datasheets show a modest improvement for higher temperatures at a few mA, but I wonder if there is more to be gained for higher currents.
In the wikipedia article on Peukert's law (which described the same capacity droop with discharge rate in lead-acid batteries), it says the drop-off law doesn't apply to lithium-ion batteries because of significant self-heating (which boosts the voltage). I wonder if the same is true for lithium primary cells?
https://en.wikipedia.org/wiki/Peukert%27s_law
The power drained from these coin cells isn't enough to cause self-heating. How much capacity can be gained by artificially heating the cell? My house is 16C in the winter, and the cells are rated up to 60C. I may try to heat them and see what happens - a little resistive heater could keep them toasty warm.
I also wonder what happens above 60C. I might fill my ammo box with sand (and maybe put it outside the house).
Are you sure? yes | no
If you are willing to work with more dangerous cells, you can abandon Li-MnO2 and go to Li-SOCl2, which brings higher capacity and currents.
One could argue that this one is still a coin cell (it's called 1/6D, but looks like a coin to me):
http://www.tadiranbat.com/assets/tl-5935.pdf
offering 1.7Ah at 3mA drain, and specified up to 10mA continuous / 50mA pulse. Interestingly, the curves show that capacity *increases* with current drain (at least up to where the curves end). Voltage is amazingly flat, and seems to stay relatively high even at high currents.
Or this one, even more coin-like (but called 1/10D):
http://www.tadiranbat.com/assets/tl-5934.pdf
These cells have a bad reputation for safety, and you'd probably want to take some serious precautions before pushing them:
https://ntrs.nasa.gov/archive/nasa/casi.ntrs.nasa.gov/19870007963.pdf
That NASA paper also has curves for discharge rates up to C/2 (850 mA for the first cell above) - see p. 5-3. They look like they can deliver a lot of power just before they explode :-)
Are you sure? yes | no
Never heard of this chemistry before. Thanks for mentioning it and for linking to the JPL reports.
Are you sure? yes | no
@Mike Szczys - no problem. You can even buy them at DigiKey. I considered them for use in TritiLEDs a while back, and have a few here, but decided the cost and safety issues weren't worth the extra capacity. CR2032s are much safer, as long as you don't eat them.
Are you sure? yes | no
Uhm, 845J at 1,5V, that doesn't look to me like it can make 10kJ you expected from nominal capacity. That looks quite horrible to me, honestly.
Are you sure? yes | no