The principle isn't that complicated. The oven consists of a container with (probably partial) insulation, a lid, and some way to heat the Galden (see https://hackaday.io/project/28449-vapsy/log/71187-basic-chamber-construction-and-heating-options for some ways of heating I have considered):
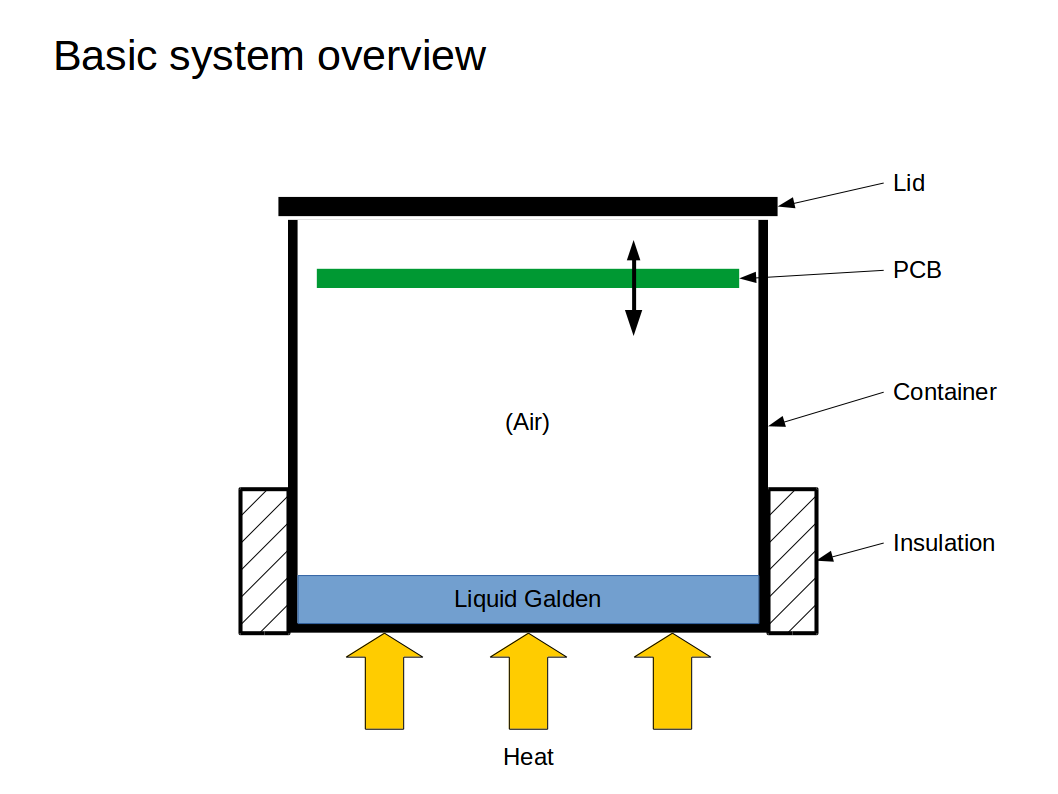
As the Galden is heated up, it will eventually reach its boiling point, start to evaporate and push out some air. Galden vapor has a higher density than air and will stay at the bottom. The lid may not seal the container because we don't want the pressure inside the chamber to increase.
Heating up and evaporating will go through multiple phases:
- plain single-phase natural convection
- "silent" boiling, with Galden evaporating at the surface (Galden/ boundary)
- nucleate boiling with vapor bubbles travelling from the bottom to the surface
- we don't want to reach the film boiling regime. Nope.
Once the second phase is reached, Galden vapor will start to move up from the surface and interact with the air above: It will mix with air, and it will push out air since the volume in the container must stay the same.
The insulation is not built up to the top of the container because we want a layer of saturated vapor (no air) above the liquid Galden. Above that saturated layer, the Galden content in the air will gradually decrease from bottom to top. This corresponds with a decreasing temperature of the Galden/air mixture. Insulating everything would result in a high vapor level, which we don't need or want.
The temperature will thus be low at the top and increase towards the bottom, up to about 230° C above the liquid. The PCB can simply be moved to the right height within the chamber to put it into an atmosphere with the right temperature along the reflow profile:
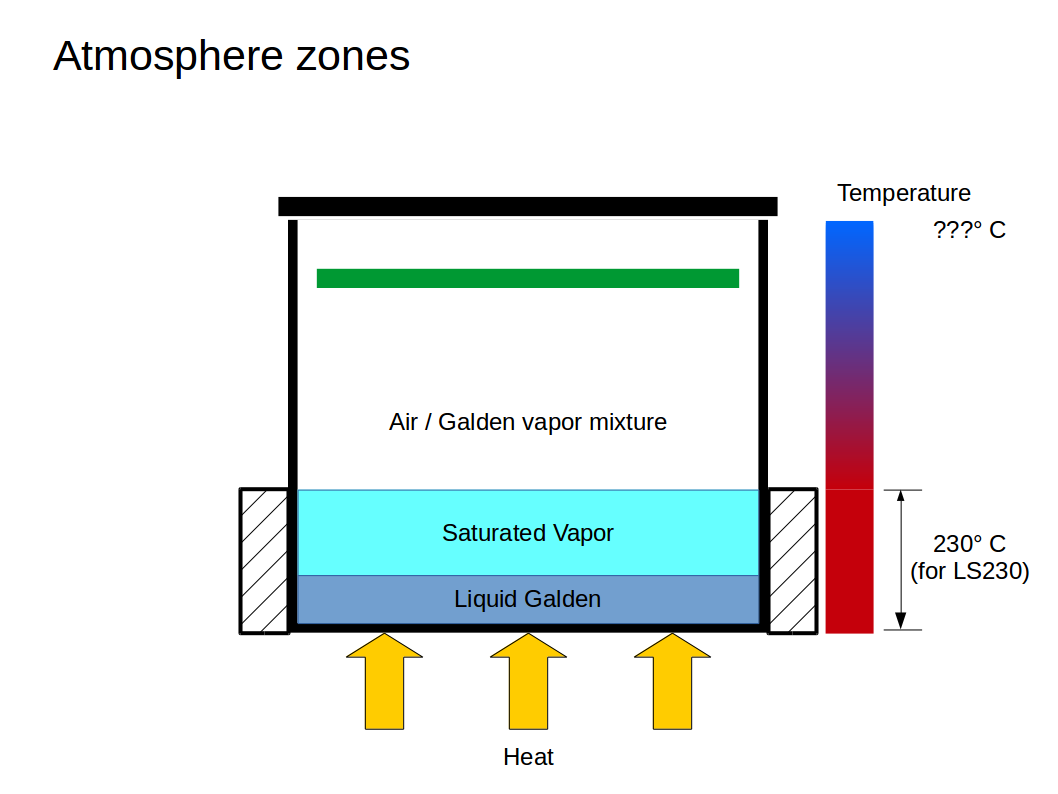
The whole thermodynamic system is quite complex and we should probably not try to control it within tight boundaries. Trying to maintain a specific temperature profile over height will be hard to impossible. Instead, the heater will be controlled to maintain a certain minimum level of saturated vapor above the liquid. Everything else will settle at some equilibrium that we just have to live with. There are two more ways to "shape" the temperature profile, though:
By increasing the height of the insulation, the saturated vapor cushion can be made higher, and the mixed zone will be shorter, probably with a higher temperature at the top. The insulation can also be thinner at the top. Choosing a higher container gives more flexibility here.
Cooling the upper part of the chamber is another way, and comes close to the opposite of pulling the insulation higher. Can also be used during cooldown. The original vapsy needed ages to cool down.
In order to dig more into the details of the various types of heat transfer involved I'll need more information about the properties of Galden. The data published by Solvay is given at 25° C, which is far from the boiling point. The surface tension for example has an influence on the heat transfer during nucleate boiling, and it will decrease with temperature. It would be plain wrong to use the 25° C value for that.
 Christoph
Christoph
Discussions
Become a Hackaday.io Member
Create an account to leave a comment. Already have an account? Log In.