Isothermal Calorimeter
This project is a Heat Conduction Isothermal Calorimeter based on the designs of Dr Lars Wadsö, Sweden.
This project is a Heat Conduction Isothermal Calorimeter based on the designs of Dr Lars Wadsö, Sweden.
To make the experience fit your profile, pick a username and tell us what interests you.
We found and based on your interests.
Hello everyone,
I am trying to organize this build in a single place on my weblog and will be updating it regularly with more diagrams and eventually some Arduino/Teensy source code for data acquisition into the SD card.
Please let me know your thoughts on this post here: https://hefnawi.me/posts/custom-built-isothermal-calorimeter/
I would like to here your feedback as well as any suggestions you would like to know or read about this build.
Thank you very much,
Ahmed Hefnawi =)
I added some NaHCO3 with water (exothermic reaction) into the sample vial and the reference vial contained sand at that time.
The following is a plot of the raw calorimeter readings in micro-Volts versus the time in seconds, the plot was created in Python using matplotlib.
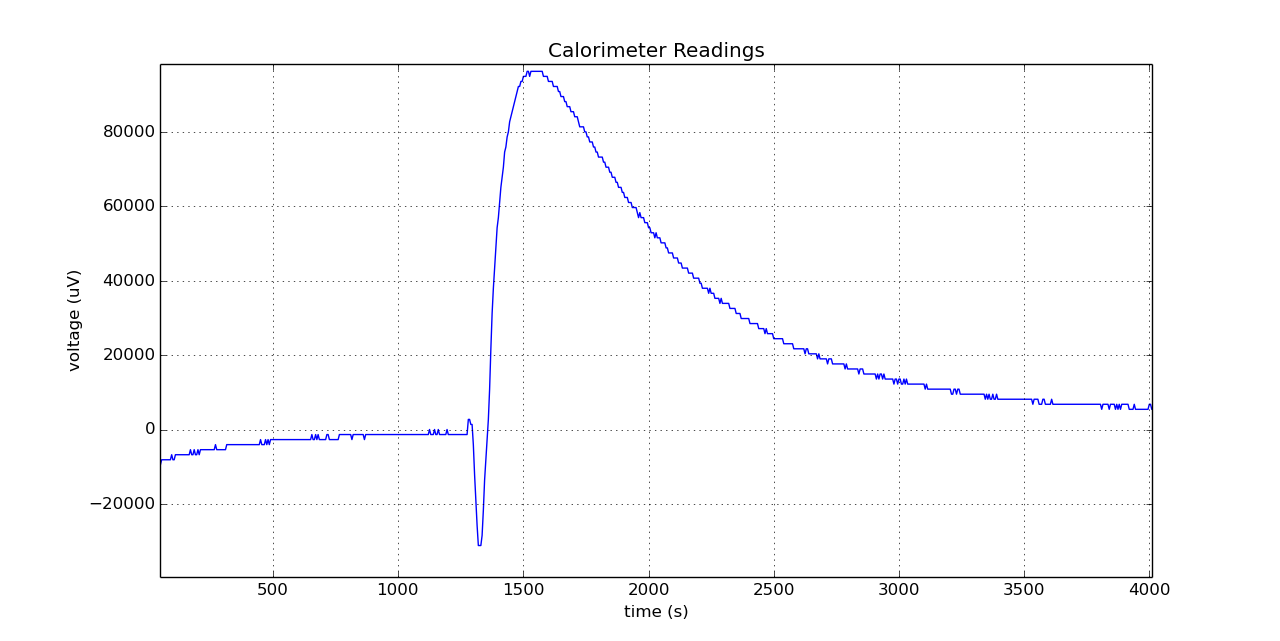
I thought I should add the thermal power after conversion as well in milli-Watts,

Calibration Coefficient as well as baseline was calculated using the calibration procedure explained in the previous log.
Next, I'll start calculating the heat of hydration for a cement sample with the help of a PhD student at my University.
Where,
U: Calorimeter Output in Volts (V)
P: Thermal Power in Watts (W)
Q: Heat Produced in Joules (J) or J/g
mc: Cement Weight in grams (g)
The duration of the experiment will be 3 days.
Also the calorimeter readings are logged every 5 seconds onto the microSD card.
It's time for calibrating the calorimeter! There are two parameters I'm interested in for the cement hydration process, which are:
I'm busy at college so didn't have the chance to update the log much further.
I created a PCB to plug-in the Teensy 2.0 board and handle external power using a single cell 3.7V LiPo 4000mAh, running the Teensy at 3V instead of 5V will consume less power and give the battery roughly 6-8 days of continuous data logging to the SD card. During the experiment we experienced periodic loss of power so that's why we are adding external power source.
Also, I believe the battery powered device will reduce lots of the noise from the AC adapters.
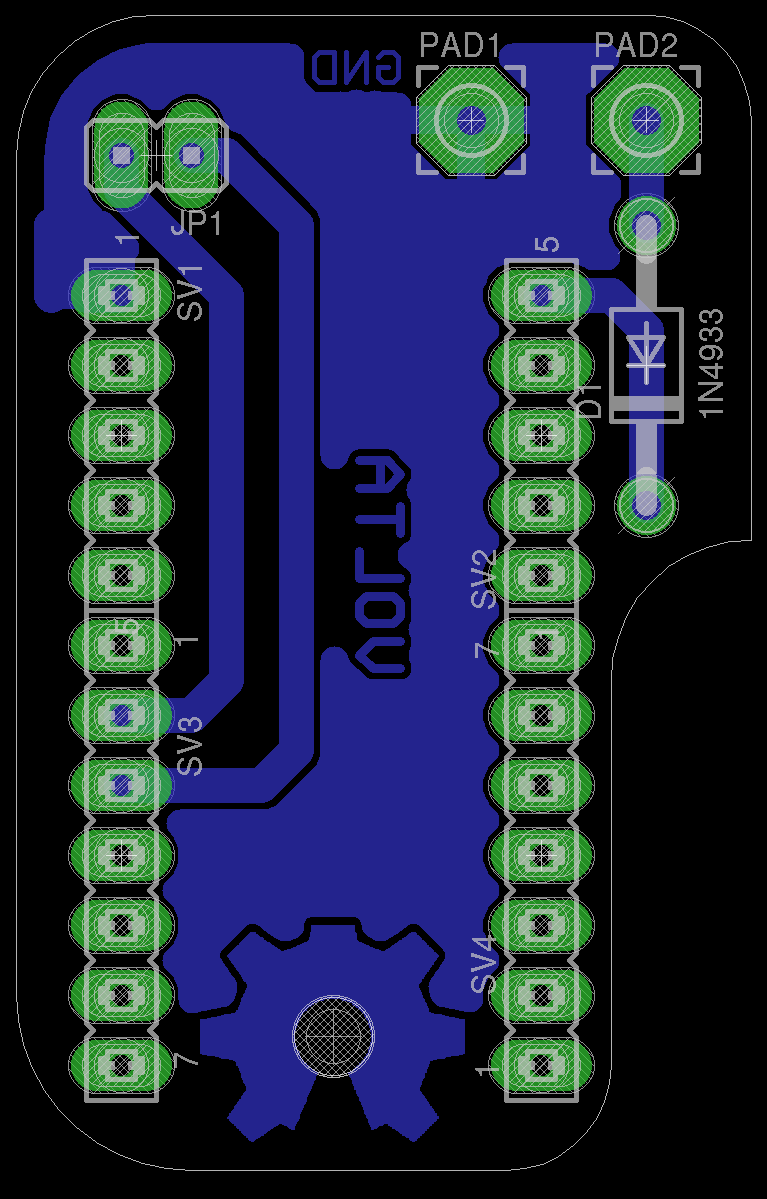
I'll be adding more photos after I finish etching the board and soldering the components.
The prototype is complete now, I calibrated my sensors with water in the reference vial so this calibration coefficient will work with solution but I will have to re-calibrate it for cement.
The calibration for cement will require filling the reference (inert) vial with Sand or NaCl. Because the specific heat capacity for water is 4182 J/kg while Concrete is 880 J/kg which matches that of Salt and NaCl of 880 J/kg. Sand will be accessible in our Lab, and its specific heat capacity is 830 J/kg.
A PhD student at my faculty is using the device now to perform the heat of hydration of cement experiment over an interval of 7 days. The data is being logged to a micro SD card into a CSV file every 5 seconds (which can be opened via libreOffice Calc or any spreadsheet program).
I'll be adding more specific details about the build and the calibration process.
Create an account to leave a comment. Already have an account? Log In.
Waakaykom Assalam bro ^^
I'm glad my project could be of any help to you. You will find the components in the details above, you can also use an Arduino instead of the Teensy 2.0, also you can exchange the ADC with any other alternatives available like the ADS1115 or others.
I will post build instructions soon along with source code and CAD drawings for the current design. Also don't forget to read the original paper in the links above: http://lup.lub.lu.se/luur/download?func=downloadFile&recordOId=1169699&fileOId=1266405
Meanwhile, if you have other questions let me know and I will try to answer them shortly =)
Ahmed
it is very kind of you my bro
would you send the building instuction of the calorimeter and its detail to my email ( agungsdjohan@gmail.com ), please?
Very cool! Looking forward to seeing results from your unit. Any chance you'll test other materials besides cement?
Thanks Zach! Actually cement will be the major test specimen, but once I get good results from the unit I'd love to test other processes as well. Which materials are you interested in?
Oh man, I dunno. Other exothermic processes, I suppose? What about one of those hot packs that works by crystallizing a supersaturated solution of some kind? http://thermo-pad.com/how.htm
No worries, first things first. I'll look into it once I get cement heat rate.
Thanks again for the suggestion.
I added some NaHCO3 mixed with H2O, will post the results shortly.
Paywalls stink. Here is the link for the open version of the paper: http://lup.lub.lu.se/luur/download?func=downloadFile&recordOId=1169699&fileOId=1266405
Become a member to follow this project and never miss any updates

 Scott
Scott
 Frédérik Berthiaume
Frédérik Berthiaume

 iSax
iSax
assalamualaikum yaa akhi, syukron for your project report
can you help me for my final project? it is about how to built a isothermal conduction calorimetry, including components and how to install it into a apparatus
i really hope for your help