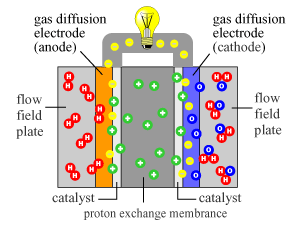
Access to readily available electricity is something, that evades much of the worlds population. While at the same time, the amount of human generated waste continues to grow exponentially. Besides waste, much of the world is still struggling to to gain access to clean water, medicine, education, and safe long term food storage. Many of theses issues could be solved, with access to basic electrical power.
In the 21st century it is virtually impossible for people in under developed areas to work toward bettering their circumstances without access to technology. Several organizations have already started to work toward increasing access to meet that needs. However, access to electricity to power the technology is again scares for many. Without electricity many people are left behind, with little hope of succeeding in a rapidly developing and globalized world.
The ultimate goal of this project is to help empower people all over the world to generate their own power, using materials that would usually be discarded with little thought or concern.
Not only am I hoping that this project will help people in underdeveloped regions, but I am also hoping it can be used to help with recovery after a natural disasters, or be applied to any number of other situations were it would be useful.
If you have any comments or suggestions, please post them. I'll never turn down free advise.
P.S. Don't forget to Follow and leave a Skull if you like what you see.
 Chali Baicunn
Chali Baicunn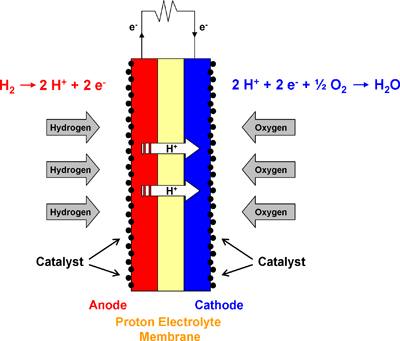











 Glytch
Glytch
 Brian Gilbert
Brian Gilbert
 sparks.ron
sparks.ron
 Sam Smith
Sam Smith
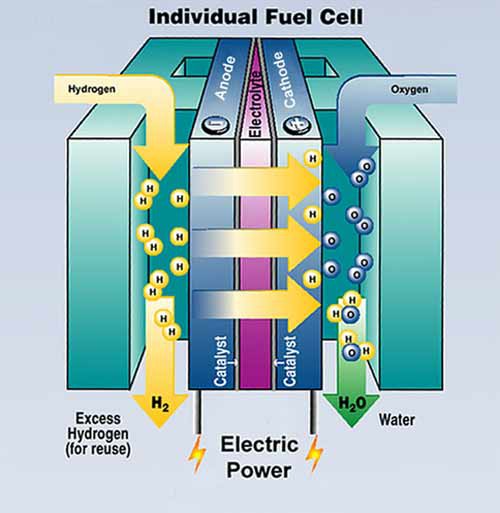
Good article on phosphoric acid electrolyte I stumbled across while looking for electrolyte concentration.
http://phys.org/news/2012-04-chemists-molecular-fuel-cell-electrolyte.html