The Metabolizer doesn't require perfectly dry material to make fuel, and in fact I've run it with some pretty seriously wet woodchips, with no problem. However, being able to remove all of the water from incoming material before it enters the reactor to be thermally decomposed would have several distinct benefits.
First of all, if you know the material is completely dry, you get a better sense of how much actual biomass you're loading in by weight, which lets you figure out how much energy you put in, versus how much energy you get out, and that lets you calculate the overall efficiency of your system.
Second of all, it ensures that the liquids that distill out aren't watered down, which means there is less refining to do, and less liquid to deal with.
Third of all, if you could remove the water in such a way that you were able to recover it, then the system would actually produce clean water, would be super neat since humans need water to live.
So I've been looking into ways to dry out the material before it goes into the metabolizer, that isn't too insanely energy intensive. A simple dehumidifier inside a closed container is remarkably viable considering you get quite a lot of very-nearly-drinkable water out, but it's still a bit too power hungry for my liking, and it requires an extra bit of specialized equipment that is prone to breaking.
So the most intriguing option I've found is using desiccants, specifically, "zeolite" desiccants, which are a particularly fascinating material and so I wanted to do a log about them.

Zeolites are a class of naturally-occurring alumino-silicate clays (although man-made zeolites also exist), that just happens to have a particular molecular structure that gives them some very interesting properties.
Zeolite means "Boiling Stone" in Latin, because when zeolite is heated to around 300-600F (depending on the type) it expels a large amount of steam- seemingly from nowhere. The reason this happens is that the crystalline structure of Zeolite acts sort of like a cage for water molecules. Water molecules are attracted by the inter-molecular forces inside the zeolites, and at room temperature water molecules migrate into the crystal matrix and just kinda get stuck there. This is called "adsorption".

There are over 50 types of zeolites, but the kind I'm looking at, 3A (which refers to their pore size- 3 angstroms across), can adsorb roughly 20% of their dry weight in water. For this reason, they are often used in industry as desiccants. They're also widely used in chemistry as "molecular sieves" since only very simple, very small molecules can enter (and get stuck in) their crystal matrix, and everything else just passes between the pellets. So they're often used to selectively remove gases such as water vapor, hydrogen sulfide, and carbon dioxide from gas streams.
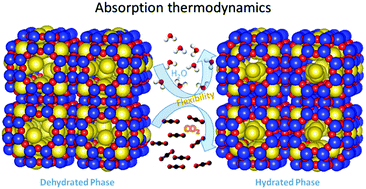
Zeolites will absorb water from air until they are saturated- and all you have to do put them in the same air-tight container with a wet material, and they will suck the moisture out of the material. Once they are saturated, they can be completely regenerated by heating them up to their recovery temperature. The heat causes the crystal lattice to expand, which eventually allows adsorbed water to escape (which is almost pure water, and easy to capture and condense).
But the most intriguing part of this thermodynamic cycle (to me, currently) is that it acts as a near-infinitely regenerative heat battery. When you heat up zeolites to their recovery temperature (which is on the lower end of the same ball park as the pyrolysis temperature of biomass- temps the metabolizer can easily reach) they start releasing water vapor, and that takes energy away from the system- it's an endothermic reaction. You have to keep adding heat to the system, but it won't get any hotter until all the water is released. You spend heat energy in order to recover the zeolite, and that makes it more efficient than using a vapor-compression dehumidifier, since you don't have to refine the fuel heat into electricity.
When dry zeolite begins to hydrate, it heats up- it's an exothermic reaction. Like....really hot. Nearly the same amount of energy is released in the hydration phase as went into the dehydration phase. And this heat actually needs to be dissipated somehow in order for the zeolite to be an effective desiccant, since the hotter it is, the less effectively it can retain water.
I've only just started experimenting with this, but my hunch/hope/suspicion is that I can fill a keg with zeolite pellets, just like I do with wood chips, use heat from charcoal to recover the water from it, potentially recover that water for later use, and then use the keg full of dry zeolite to dry kegs full of biomass. Most raw wood chips have a water content of around 20%. Zeolites have a water capacity of roughly 20%.
A 5 gallon keg full of dry zeolite (which would cost around $100) should be in the right ballpark to suck very nearly all the moisture out of a 5 gallon keg of wet biomass. If you put the kegs side by side, and insulated them, as the zeolite keg heats up from the heat of adsorption, the biomass in the other keg would warm up via conduction, causing the air inside to warm up as well. Warm air can carry more moisture, and that warm wet air would migrate due to the pressure gradient to the zeolite, accelerating hydration, and increasing the heat, until the material is completely desiccated. If that's the case, the zeolite kegs could be used as a sort of "load dump" and heat battery, that has the additional benefit of recovering clean water.
Right now, I only have about a pound of zeolite to test with, but I'm really intrigued by the possibility of such a simple, stable, heat-driven desiccation system!
 Sam Smith
Sam Smith
Discussions
Become a Hackaday.io Member
Create an account to leave a comment. Already have an account? Log In.
As always: super interesting read! Enjoyed reading it, same with the log about "what is plastic"!
Are you sure? yes | no