July 9, 2018
What's the point of having a new toy if you don't play with it?
First thing I did was to replace the J-hook test leads with 22 gauge wire soldered onto the plated-through-holes at the board edge. A bit of heat shrink and I have a nice rigid cable that I can plug into my breadboard. Much cleaner and easier to use now, although not as rigid as I would like. The third hand as a support isn't much better, so I need to develop some way to mount the board in a more reproducible manner. Maybe using the mounting holes...


I will turn the spectrometer on the multi-colored paper targets in a bit, but first, let's take a look at some simple organic objects; an apple, a banana, and my palm:

What color is the apple? Banana? Well, I see yellow and green in both, and a bit of red in the apple. What color is my palm? It is somewhat apple-like, but not green ;> What does the spectrometer say?
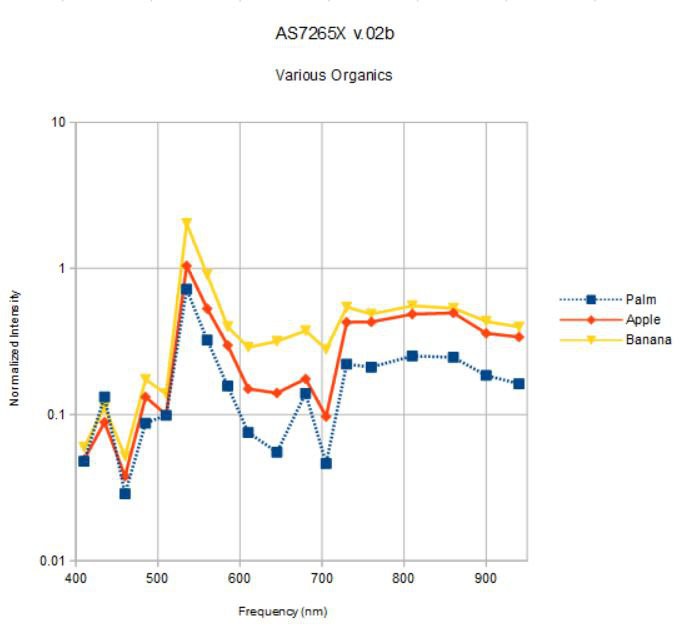
I am plotting the spectrum measured from each object at 16x gain, 100 ms integration time, and 25 mA for both white and IR broadband source current. The spectra are normalized by the spectrum measured under the same conditions for the vanity mirror as reference. I expected the spectra to be less than one (less reflective than a silvered mirror) but I didn't control the distance from the spectrometer to the objects very well; hard to do with curved surfaces in general.
Maybe my palm is green! The peak at 535 nm dominates for all three objects. None have a significant blue component and above 600 nm the banana, somewhat surprisingly, has the flattest spectrum with the highest reflectivity. I am not quite sure what to make of this. Are the apparent color differences we see with our eyes in the objects above really due to small, relative differences between similar spectra? What happens when we use obviously different objects, like the colored paper above?
I repeated the same experiment (with the same settings) as above but this time I took more pains to keep the distance between the spectrometer and objects the same by leaning the paper targets agains the mirror surface and restarting the sketch to grab a new spectrum. It was not possible to keep the paper targets completely flat nor in the exact same position, still I was surprised that in some cases I measured twice the light (twice the calibrated intensity anyway) reflected from the paper target than from the silvered mirror!
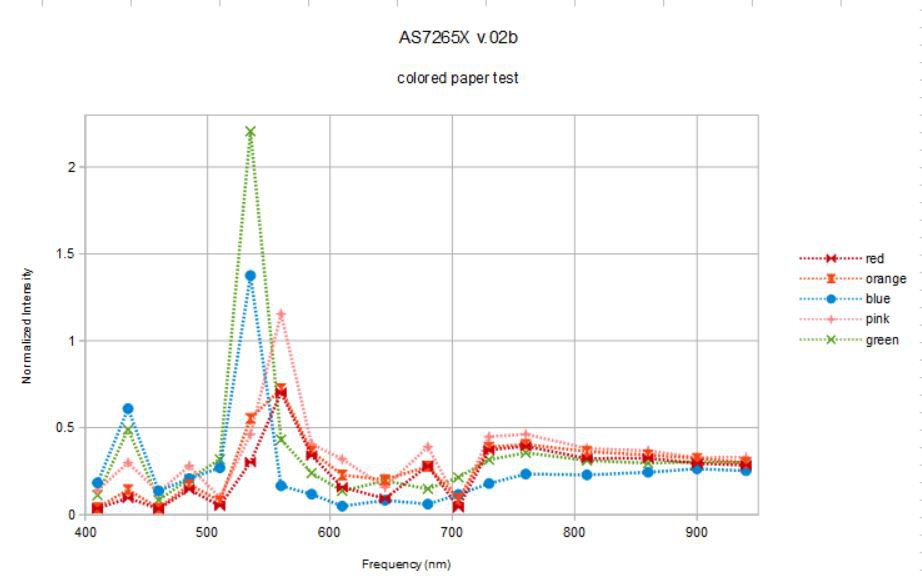
Well... this is sort of psychodelic, flourescent colored paper. I wonder if the dyes that give the paper these colors are really flourescing under the broad-band illumination? This wouldn't be too surprising given the very strong deep blue (410 nm) component of the source light. Do the spectra make sense?
The blue and green paper targets reflect very little red spectral components and the orange and red paper targets reflect little blue. So this makes sense. The green and blue paper targets have reflectance peaks at 435 and 535 nm, the blue paper with a bit more 435 nm and a bit less 535 nm, green vice versa. So this makes sense. Pink, orange, and red paper targets have a dominant peak at 560 nm. There isn't a lot of difference between these, similar to the case of the organic targets above. Perhaps if we normalize everything to 1 we will discern more:

OK, the orange paper target reflectance has large components at 535 and 645 nm that the red and pink targets do not. Pink and red target paper reflectivity are almost identical below 700 nm except that the pink paper target reflects a bit more blue (435 nm) and a bit less orange (585 nm) color. Is this enough to cause the apparent color difference between this red and pink paper?
None of these tests is particularly scientific nor are they meant to qualify the photospectrometer for any particular use. It's just part of the natural process of figuring out what it can and cannot do.
One thing I have learned for sure; it is a lot of fun to use!
Just a note on cost and availablility. The ten each of the AS72651/2/3 I bought from the AMS web store for $4.50 each in May are now $6.88 each for orders of 100. Other retail sites have similarly elevated prices but little or no stock. In fact, I can't find the AS72653 in stock anywhere, even on the AMS site. The good news (maybe) is that FutureElectronics.com seems to have two of the three in stock and for $4.88 each in units of 100. So with the SJAS110100 switch (~$1), Luxeon-Z-ES broad-band led (~$1), SPI flash (~$1), pcb (~$1), and three smart spectral sensors (~$15) the total cost (with taxes and shipping, etc) of the 18-channel photospectrometer will be right around ~$20, maybe ~$25 if I include the OSRAM broad-band IR led. The cost of the first 100 in an initial production run will be a little higher since the fab always charges an NRE of $350 for new designs. So it looks like the final (I hope) design can be produced close to the $25 target cost. This means I will likely have to sell these on Tindie for $49.95, maybe a bit more.
Open source means everything about the design is (or will be) made available so you can build your own. But my Tindie store is a business and I have to make a profit. When I do have these in stock and for sale, perhaps in August, I hope you will all consider buying one.
 Kris Winer
Kris Winer
Discussions
Become a Hackaday.io Member
Create an account to leave a comment. Already have an account? Log In.