Every click-baity video starts with the final result, so here goes (not my achievement!):

I know ... everybody loves (deep) reactive ion etching with CF4 / SF6 / CH2F2 / Cl2 plasma and cheap MEMS devices...

https://www.memsnet.org/links/470/
http://www.eng.utah.edu/~gale/mems/Lecture%2008%20Dry%20Etching.pdf
RIE isn't closed-loop and ultimately it's practically impossible (think: SEM + Cl2 gas jet, super slow etch rates, https://aip.scitation.org/doi/abs/10.1063/1.3525587 ) to make plasma etching a maskless process. I could ignore shallow surface damage and ion implantation for my purposes but I like to avoid it if possible. Here's what that looks like:
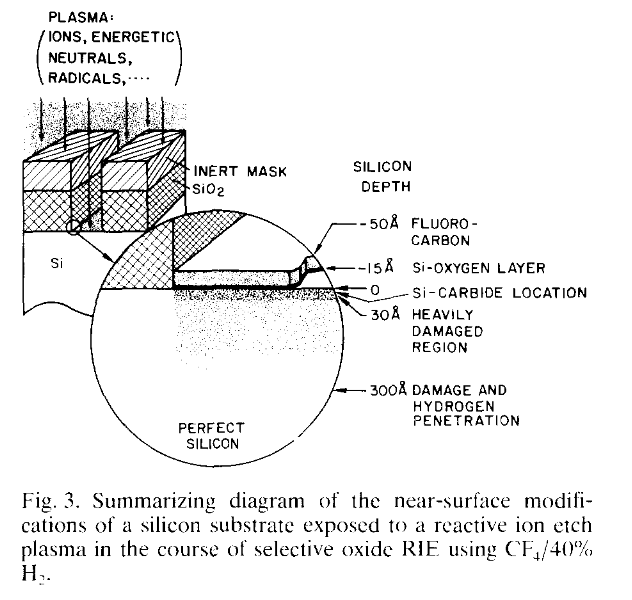
https://www.sciencedirect.com/science/article/pii/0921510789902845
Chlorine on its own or in Ar has a tendency to photo-dissociate at visible wavelengths:
Here's the absorption cross section of Cl2 gas with an onset compatible with 405nm laser diodes and a maximum that's screaming "homebrew TE nitrogen laser":
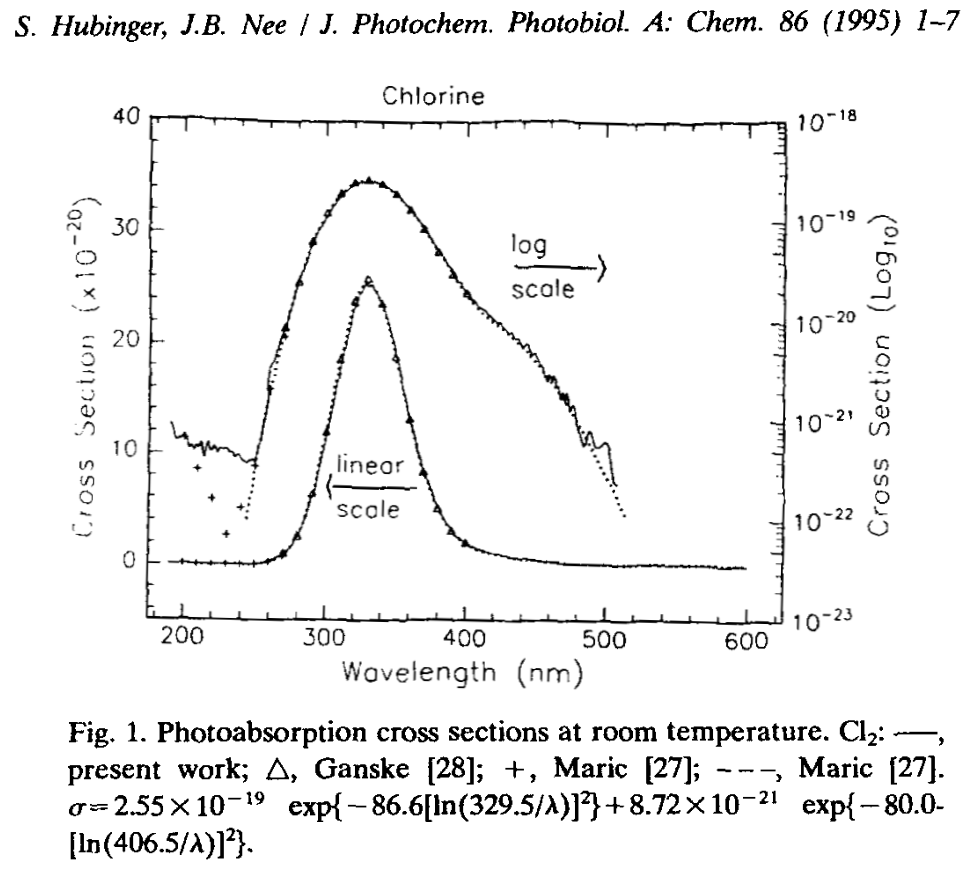
With a bond dissociation energy of 242.58kJ/mol (2.515 eV/bond) plus (I'm guessing) some additional energy to end up in an excited state the molecular absorption peaks in the UV, capable of causing localized generation of chlorine radicals.
Our goal here is to generate volatile silicon tetrachloride (cue youtube video from NileRed on its conventional synthesis with heated silicon). For this to happen, chlorine radicals must be generated in the vicinity or on the surface of the silicon specimen to be etched and be willing to react with the silicon atoms presented at the wafer surface.
Seriously, Si has to react with Cl. radicals or worse... and there's where the problems start.
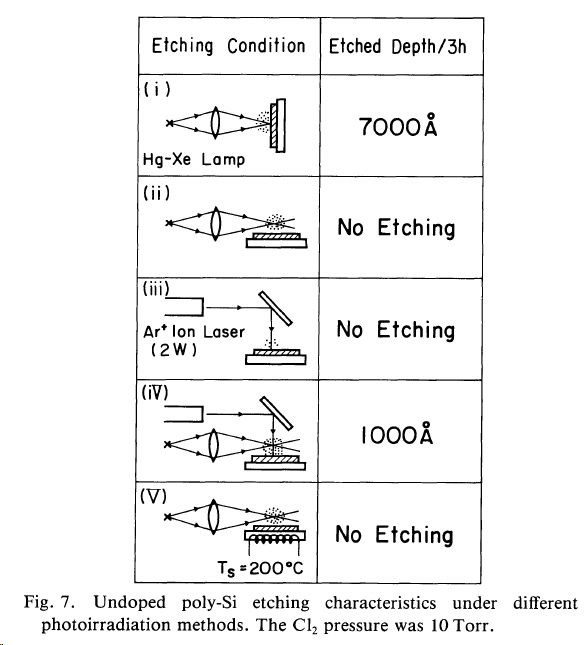
http://iopscience.iop.org/article/10.1143/JJAP.24.68/meta
"It was found that n+ poly-Si is etched by chemical reaction with Cl radicals photodissociated in the gas phase, while undoped and p+ poly-Si cannot be etched without irradiation by UV light. The primary effect of the photoirradiation is to produce electrons arising from electron-hole pair generation. The etch rates, etched features and etching products depend strongly on the electron concentration in the conduction band. The experimental results are explained by assuming that electron-attached Cl- ions penetrate into the Si lattice."
Since I'm interested in selective backthinning of p- substrates this is not going to be as easy as swapping the samples holder for a gas tight process chamber and iterating between surface profile measurements and exposure phases to achieve a controlled 3D profile.
It's not as bleak as it might look from the 1985 point of view. in fact, Vakanas 2002 states (also refering to the patent below),

https://patents.justia.com/patent/8173552
fast forwarding a few years there appears a behemoth called the VarioEdit
(as seen in Henderson 2013 https://linkinghub.elsevier.com/retrieve/pii/S0026271413001893 and https://www.varioscale.com/varioedit ):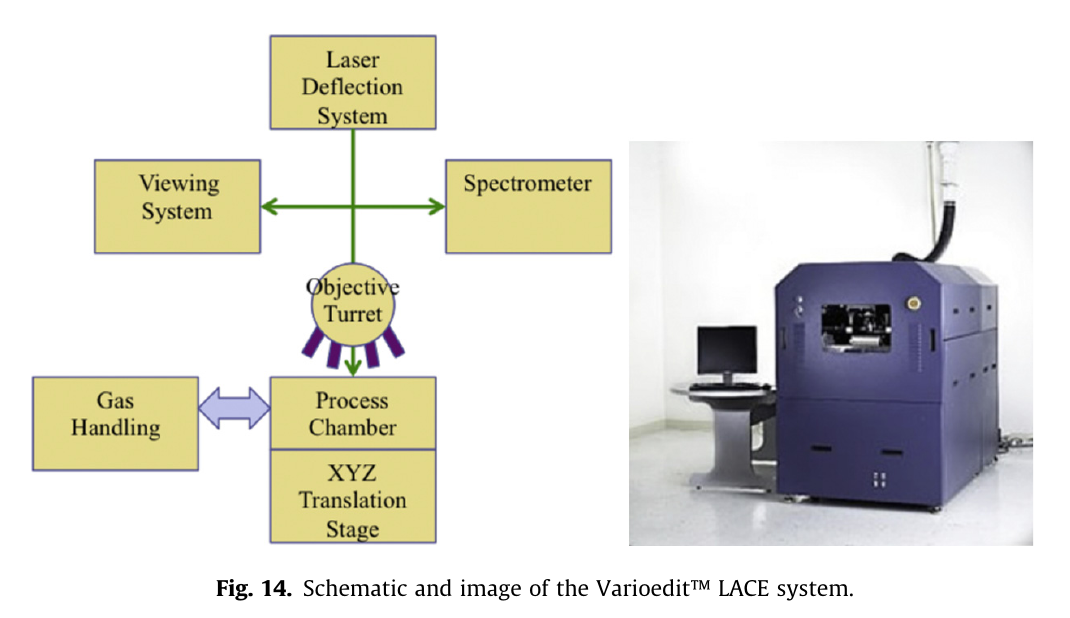
There's a project which matches the capabilities of VarioEdit:
"Intel plans to ship 10 nm integrated circuits in 2017[3], and TSMC plans to offer 7 nm chips in 2017. [4] Manufacturing at these technology nodes will require high-speed and high-resolution image acquisition for process verification and failure analysis.
The RAVEN program is focused on developing an analysis tool capable of imaging minimum size circuit features on a silicon integrated circuit chip."
https://www.iarpa.gov/index.php/research-programs/raven/raven-baa
"Officials of the U.S. Air Force Research Laboratory at Wright-Patterson Air Force Base, Ohio, announced a $23.8 million contract to Varioscale earlier this month for the Rapid Analysis of Various Emerging Nanoelectronics (RAVEN) project."
http://www.militaryaerospace.com/articles/2016/11/integrated-circuits-imaging-process-verification.html
ok, enough with the tinfoil hat stuff, back to facts from the 2002 Vakanas paper:
But now you've seen the name and recognize it on this lecture slide.

Note how the 514nm (guess: Ar* laser) photon energy is below the 493nm resulting in photodissociation of chlorine... Wonder what a few 455nm diodes could do instead, avoiding the bulky and inefficient Ar* light source.
I doubt the process relies upon heating the Si locally until the reaction to SiCl4 just takes place since it is claimed that the properties of active circuitry are not changed even when gainig backside access up to 2-3 micrometers away from transistor structures. Excessive heating would drive dopant diffusion and break functionality. Heating to 150-250°C might be beneficial though, if only for chlorine or chloride diffusion into Si.
ps. as for the electron generation / injection ... I suspect Lenard tubes are a really bad idea :)
 helge
helge
Discussions
Become a Hackaday.io Member
Create an account to leave a comment. Already have an account? Log In.
Is this project still active? What is the motivation of this project?
Are you sure? yes | no
sorry for missing your comment. Maskless lithography is still going, and I'm waiting for parts to give LACE a shot someday with beefy laser diodes (not necessarily with Ar*).
Are you sure? yes | no